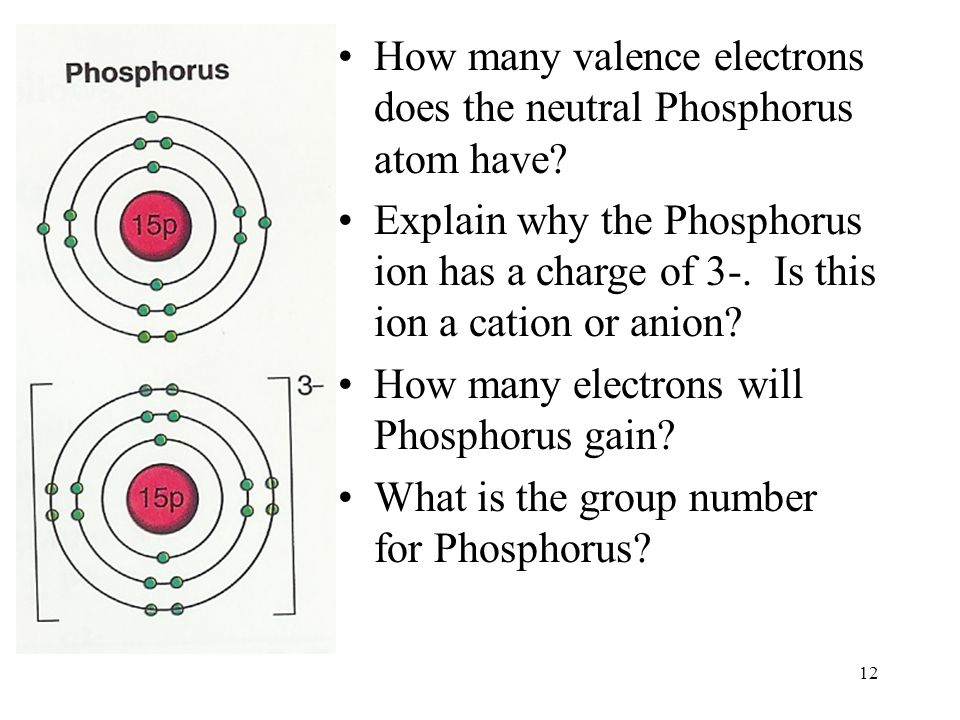 
They are both contained within the 1s atomic orbital. The electrons of any one helium atom both occupy the same atomic orbital. The Pauli exclusion principle can be more easily understood if we consider the electrons of a single helium atom. The Pauli exclusion principle states that no two electrons in any one atom or ion can have the same set of four quantum numbers ( □, □, □ , and □ ). This line of reasoning can be extended to determine that any one d orbital can only hold up to ten electrons. It also means that any one p subshell can only hold up to six electrons because p subshells have three orbitals and 2 × 3 = 6. This means that any one s subshell can only hold up to two electrons because s subshells have one orbital and 2 × 1 = 2. The total number of orbitals per subshell can be determined with 2 □ + 1 formula.Īny one atomic orbital can only contain two electrons. The f subshell has seven atomic orbitals. The s subshell has one atomic orbital, and the p and d subshells have three and five atomic orbitals. The magnetic quantum number can be any integer that ranges from − □ to + □. It also determines their orientations in space. The magnetic quantum number ( □ ) determines how many orbitals there are in each subshell. These sentences can alternatively be expressed with the following statement: s < p < d < f. The d subshell is the third-lowest-energy subshell, and the f subshell is the fourth-lowest-energy subshell. The s subshell is the lowest-energy subshell, and the p subshell is the second-lowest-energy subshell. There is a slight difference in energy between the subshells of a given energy level. The first four lowest □-value subshells can be classified as follows. The fourth electron shell ( □ = 4) can have four subshells with subsidiary quantum numbers that range from zero to three. The third electron shell ( □ = 3) can have three subshells with subsidiary quantum numbers that range from zero to two. The first electron shell ( □ = 1) can have one subshell with a subsidiary quantum number of zero, and the second electron shell ( □ = 2) can have two subshells with subsidiary quantum numbers of zero and one. This means that each energy shell can contain a number of subshells equal to its principal quantum number. The subsidiary quantum number can be any integer between 0 and □ − 1. The subsidiary quantum number ( □) determines the shape of an atomic orbital. Most chemistry textbooks focus on the first four electron shells because there are plenty of elements that have completely full K, L, M, and N shells. The atom would be extremely unstable, and it would disintegrate almost as soon as it is formed. It is incredibly challenging to produce an atom that has a completely full O shell because this atom would have a very high atomic number. Atoms tend to be extremely unstable when they have high atomic numbers. The periodic table does not include any one element that has an O shell ( □ = 5) that is completely filled with electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
